Acute kidney injury (AKI) is defined as a condition in which the kidney function and structure are suddenly affected. It is a serious disease but with a timely treatment can be reversible. AKI is common complication with critically ill patients and it is also recognized as a lifethreatening pathology closely associated with metabolic syndrome and cardiovascular diseases. AKI can occur in a variety of clinical settings and is common complication in patients after cardiac surgery.
Currently, serum creatinine is measured to diagnose AKI as it provides information of the glomerular filtration rate. However, serum creatinine levels do not change until ~50% of kidney function is lost. Furthermore, its inter-individual variability is high. This makes it a suboptimal biomarker of AKI in which the early diagnosis can significantly improve the outcome.
KIM-1 as a diagnostic biomarker
Kidney injury molecule-1 (KIM-1) has been suggested as one of the biomarkers that could improve the early diagnosis of AKI. KIM-1 levels are undetectable in normal kidneys, whereas elevated levels of KIM-1 expression was detected in the ischemic kidney in an animal model of disease, as well as in humans. In urine, KIM-1 concentration was shown to increase up to 3-7 ng/ml from its normal concentration of less than 1 ng/ml following an ischemic kidney injury. KIM-1 levels begun to increase as early as 6 hours after an ischemic insult and remained elevated for a period of 48 hours post-injury.
In addition, KIM-1 has been suggested to have predictive value for AKI in patients undergoing cardiac surgery. It might also be utilized as a nephrotoxicity biomarker in preclinical studies of drug candidates since kidney tissue may suffer from ischemia as a result of drug-related response. The Food and Drug Administration has recognized KIM-1 as an appropriate biomarker for renal injury in preclinical studies of pharmacologic agents. According to some publications, KIM-1 could also be used for detecting certain types of cancer.
Hytest provides two monoclonal antibodies (MAbs) specific to the ectodomain of human KIM-1. They are suitable for a quantitative detection of KIM-1 in urine using a sandwich ELISA.

Quantitative sandwich immunoassay
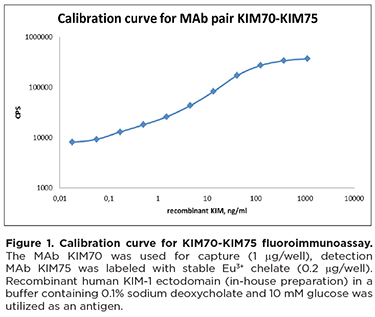
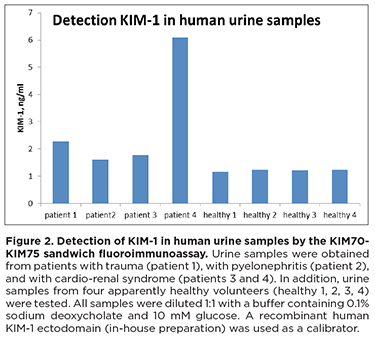
Calibration curve for KIM70-KIM75 (capture-detection) is shown in Figure 1. The limit of detectionfor this pair in fluoroimmunoassay is ~0.2 ng/ml. The prototype assay was also tested for its ability to detect native KIM-1 in urine samples from patients with cardio-renal syndrome, trauma, and pyelonephritis (Figure 2).